Linking Parkinson's Disease and Aging
Epigenetic Analyses of the Substantia Nigra Provide New Insight into Links Between Aging and Neurodegeneration: A new study integrating epigenetic and transcriptomic data from the aging mouse substantia nigra provides insights into the link between aging and Parkinson's disease.
Why Does Increased Age Represent a Risk for the Development of Parkinson's Disease
The progressive loss of dopaminergic neurons in the substantia nigra represents the underlying cause of the motor symptoms associated with the development of Parkinson's disease. Links between normal aging and the development of this prevalent neurodegenerative disease appear at multiple levels: Parkinson's disease mainly affects individuals aged over sixty, increasing age constitutes the predominant risk factor for disease development (Hou et al.), and several aging-associated cellular alterations mirror disease-associated pathological processes (Hindle 2010, Reeve et al., and Collier et al.). A 2024 study that employed single-cell transcriptomic and epigenetic analyses defined the cellular and molecular changes associated with aging and Parkinson's disease development in human samples (Adams et al.). While this research provided much-needed insight into potential mechanisms linking aging and Parkinson´s disease, the use of human cells complicated the interpretation of age-related effects. For this reason, researchers led by Bing Ren (University of California, San Diego) recently performed single-nucleus multiome sequencing on over 40,000 cells from mouse substantia nigra samples at multiple points during aging to support the unbiased assessment of cell-type-specific transcriptional and epigenetic alterations across a lifespan (Wang et al.). Their recent Genome Research study now reports a single-cell multiomic atlas of the aging substantia nigra, which may deepen understanding of the links between aging and Parkinson's disease development and, as such, help to develop preventive and therapeutic approaches to this devastating and unfortunately prevalent neurodegenerative disease.
Paired-Tag technology from Epigenome Technologies generates joint epigenetic and transcriptomic profiles at single-cell resolution and detects histone modifications and RNA transcripts in nuclei with efficiency comparable to single-nucleus RNA-seq/ChIP-seq assays. Could the application of Paired-Tag to single cells from the aging mouse substantia nigra have provided deeper epigenetic insight into the links between aging and Parkinson's disease pathogenesis?
Integrated Epigenetic and Transcriptomic Analyses Link Aging and Parkinson's Disease
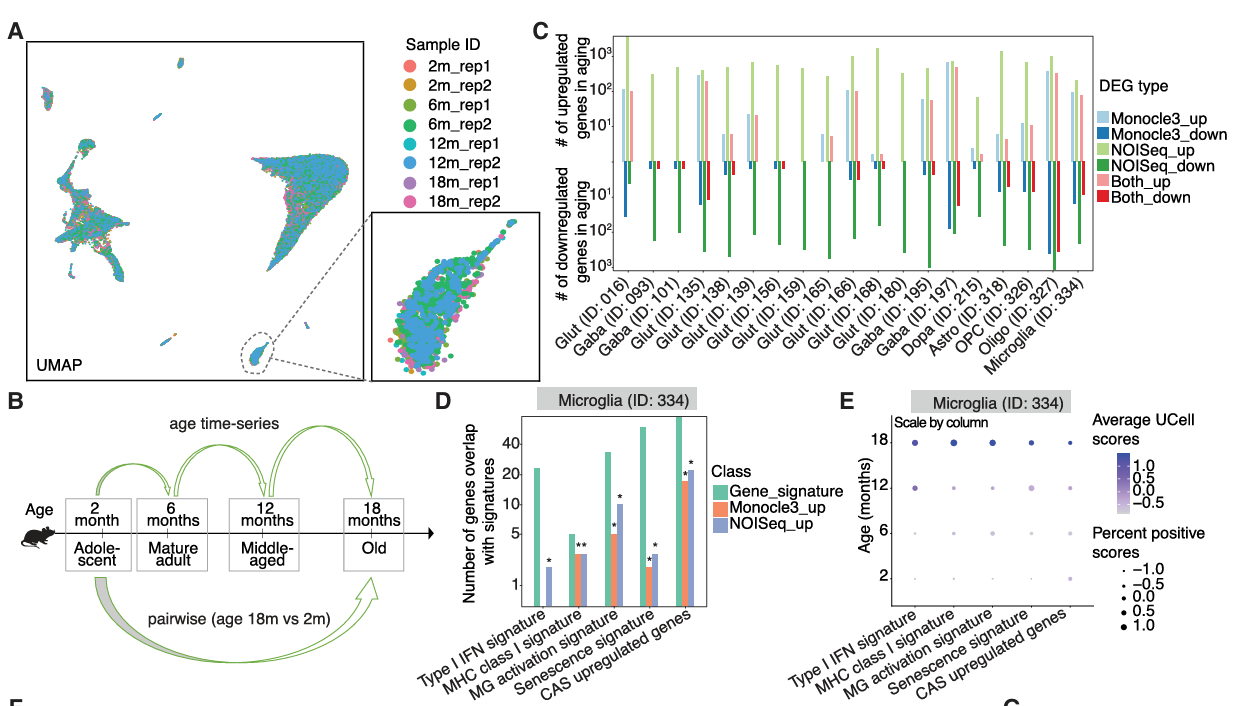
To investigate cell-type-specific transcriptomic and epigenetic changes occurring in the aging mouse substantia nigra, the authors generated single-nucleus (sn)ATAC-seq and snRNA-seq profiles for this brain region in mice at 2-18 months - covering adolescence, adult maturity, middle-age, and advanced age - constituting a practical model for studying aging-related diseases. Analyses of these datasets created a single-cell multiomic atlas of the aging mouse substantia nigra comprising ten cell classes and twenty-seven cell subclasses; importantly, initial analyses revealed that aging did not induce significant alterations in the cellular composition of this all-important brain region, although they did observe significant alterations in gene expression and chromatin accessibility in multiple cell types.
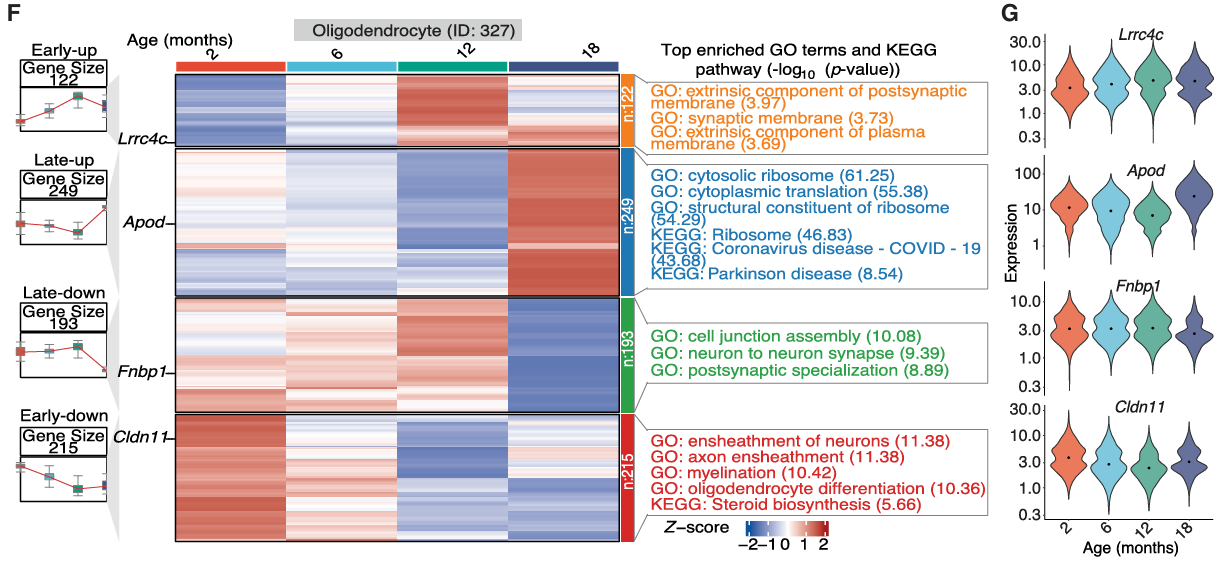
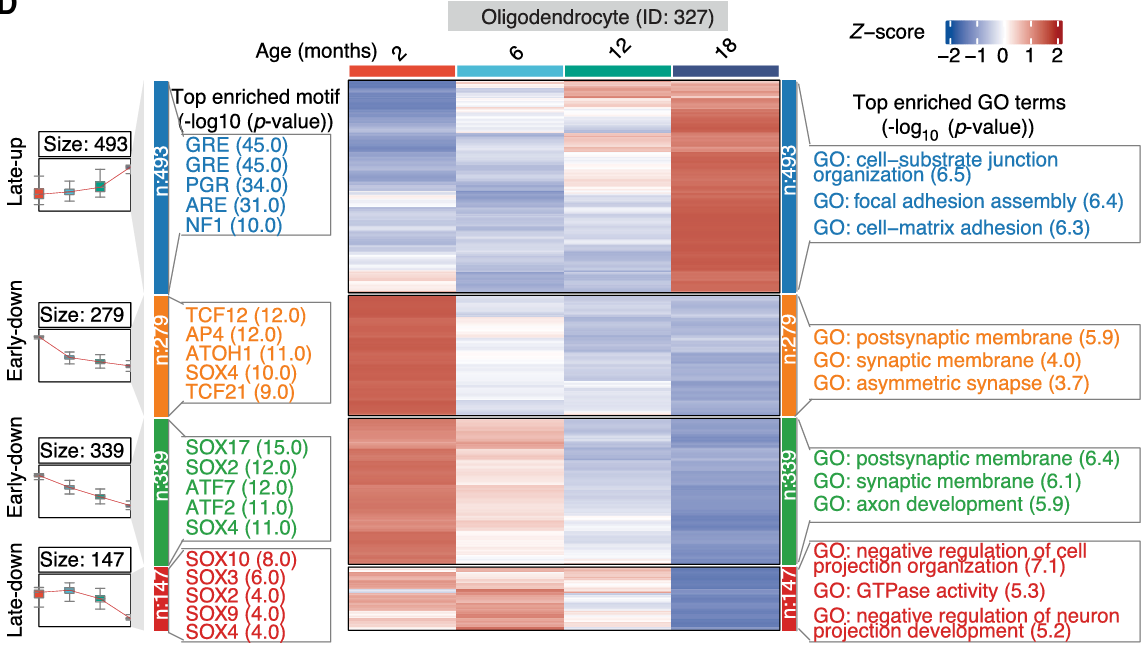
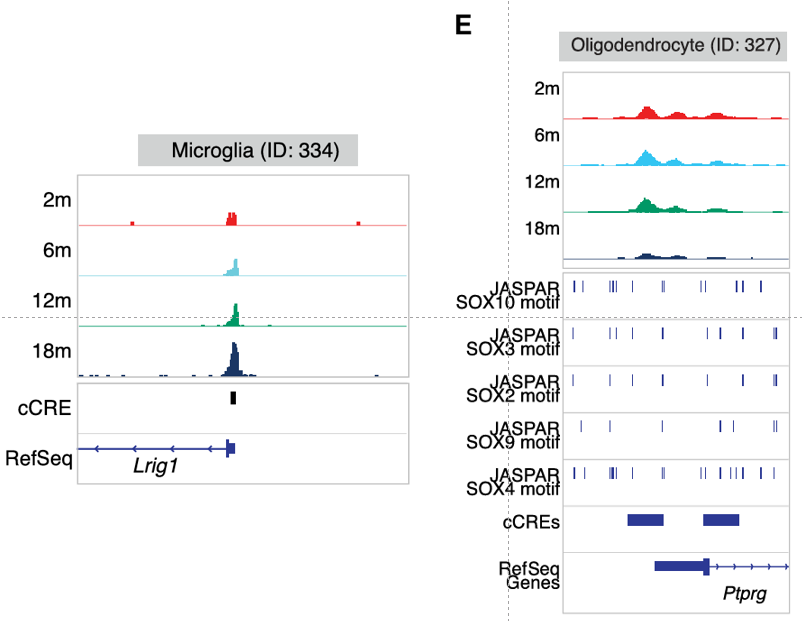
Importantly, deeper analysis of this epigenetic and transcriptomic dataset helped to uncover age-associated changes occurring at the cell-type-specific level. First, the authors discovered the increased implication of two canonical hallmarks of aging - inflammation and cellular senescence - in microglia within the substantia nigra during the aging process, which may contribute to the increased vulnerability to neurodegenerative diseases. Secondly, this analysis revealed that age-associated differentially expressed genes across several cell subclasses formed part of Parkinson´s disease-relevant pathways, including a set of genes whose expression increased in oligodendrocytes (the most abundant cell type in the substantia nigra) at later stages of aging. The multiomic approach taken by the team supported the identification of regulatory changes during aging in oligodendrocytes, including alterations in the Sox family of transcription factors, which remain critical for neurogenesis and whose dysregulation has been linked to Parkinson's disease (Stevanovic et al.). In addition, linking age-associated differentially accessible regions to differentially expressed genes in specific cell types provided a deeper understanding of the dynamic nature of cellular regulatory networks in aging that may support Parkinson's disease progression. Of note, dopaminergic neurons (which display specific vulnerability in Parkinson's disease) displayed the increased expression of genes associated with neurodegenerative disease pathways with increasing age and the decreased expression of genes associated with cell adhesion pathways.
The authors next integrated the mouse aging datasets described above with published single-cell RNA-seq datasets from human patients to pinpoint cell-type-specific genes affected by Parkinson's disease and aging. Their analysis revealed eighty-five genes that consistently exhibited differential expression with aging and during Parkinson's disease development. Specific examples with links to the development of Parkinson's disease included: i) the heat-shock protein genes Hsp90aa1 and Hsp90ab1, which underwent upregulated expression at late stages of aging in oligodendrocytes, microglia, and glutamatergic neurons, ii) Apoe and protein folding-related genes, which underwent upregulated expression at late stages of aging in oligodendrocytes, and iii) myelination-associated genes, which underwent downregulated expression at early stages of aging in oligodendrocytes. The authors suggest that such stage-specific alterations reflect proactive and reactive responses during aging and pathological processes, and that age- and disease-associated alterations in gene expression reflect molecular changes that predispose the aging brain to neurodegenerative disease.
New Insight into the Pathogenesis of Parkinson's Disease
Overall, this exciting multiomic study of the aging mouse substantia nigra highlights: i) significant alterations in gene expression and chromatin accessibility in multiple cell types; ii) the association of genes whose expression increases with aging with Parkinson's disease-related pathways; and iii) age-related changes in the gene regulatory networks. Overall, these findings provide valuable insights into the cellular and molecular mechanisms underlying the increased risk of Parkinson's disease with age, and the resultant multiomic cell atlas of the aging substantia nigra may serve as a highly useful platform for further insights into Parkinson's disease-associated alterations.
The implementation of Paired-Tag technology from Epigenome Technologies, which generates joint epigenetic and transcriptomic profiles at single-cell resolution and detects histone modifications and RNA transcripts in individual nuclei with efficiency comparable to single-nucleus RNA-seq/ChIP-seq assays, has the potential to provide deeper insight into such research aims. What more could the simultaneous single-cell analysis of histone modification and transcriptomic profiles tell us about the links between aging and Parkinson's disease in the substantia nigra?